There are two main routes to human longevity: slowing down the aging process and reversing it. So far, the first strategy has been clearly dominant. Obviously, it is conceptually easier to find ideas to slow down the onset of age-related dysfunctions (for example, by targeting metabolism) than ways to rejuvenate organisms (i.e., converting them from an older state to a younger one).
The most significant risk factor for human mortality, aging leads to functional decline, increased frailty, and increased susceptibility to chronic diseases (Brett & Rando, 2014; Lopez-Otin et al., 2013) Current strategies for life extension can be divided into three main categories: a) those that treat the direct causes of mortality, b) those that delay or attenuate the biological aging process and c) those that achieve rejuvenation (i.e., the reversal of aging).
At Ourenstividad we emphasize the second point due to our local diet with important antioxidants, our environment and our healthy habits.
We know that most neurons differentiate terminally during development, remain in the body throughout the life of the organism, and cannot be replaced naturally. However curiously in some young species certain parts of the body or organs can regenerate or grow back into an almost identical structure of the lost tissue.
At the molecular level, several omics-based approaches have emerged to quantify rejuvenation and aging. It is possible to assess biological age through biological aging clocks that focus on the epigenome, transcriptome and immunome or epigenetic aging clocks based on DNA methylation whose epigenetic predictions reflect various life-extending treatments, such as calorie restriction and elimination of the growth hormone receptor. (petkovichet al., 2017).
Today we can know the biological age and it is known that . Epigenetic age acceleration was associated with many age-related conditions, such as all-cause mortality, cognitive performance, frailty, progeria, Parkinson’s disease, Werner syndrome and Hutchinson Gilford progeria syndrome (Breitling et al., 2016; Horvath et al., 2018; Horvath & Raj, 2018; Lin et al., 2016; Maierhofer et al., 2017; Marioni, Shah, McRae, Chen, et al., 2015; Marioni , Shah, McRae, Ritchie, et al., 2015). Together, these studies offer evidence that aging clocks based on methylation levels can accurately track biological age and act on future rejuvenation therapies (already being successfully experimented in mice), with genetic remodeling playing an important role. (Horvath , 2013; Petkovich et al., 2017).
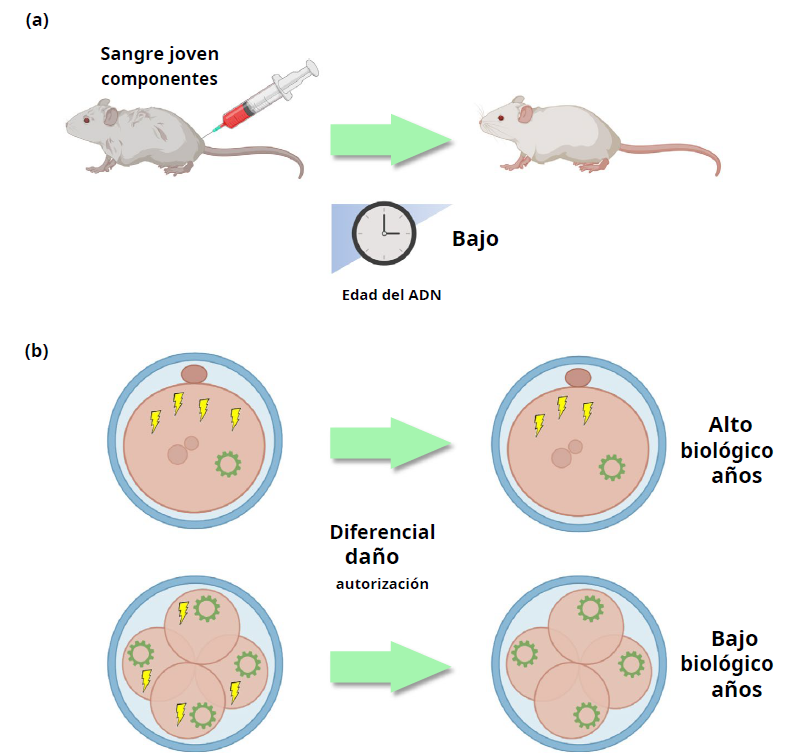
Regarding rejuvenation, there are two emerging theories, such as heterochronic transplantation based on connecting the young circulatory system with an old one. Studies carried out on mice showed that they experienced the presence of youthful characteristics in the brain, muscles and liver, characterized by greater cognitive function. In these cases, with bone marrow transplantation, the epigenetic age of the blood of the recipients coincides with that of the donors, although the systemic effect could not be established. Also important in this process is the elimination of biochemical damage (something that certain dietary behaviors help us with), which is carried out incompletely through processes such as DNA repair and detoxification pathways at the cellular level as important as autophagy. at the subcellular level and senescence/apoptosis at the cellular level.
Rejuvenation can also be focused from the point of view of DNA methylation and cumulative damage, and can act at the level of rejuvenation in embryogenesis. The mechanism would be due to “dilution of harmful molecules” within the cells. Thus reducing the biological quality of the embryo. The recent development of the first single-cell epigenetic clock, ESCAÑA, helped increase the resolution of the natural aging process (Trapp et al, 2021)
By removing damage to the stem cells of mice in culture, early biological rejuvenation is initiated to begin aging at a lower biological age, successfully prolonging lifespan.
Therefore, the two mechanisms of rejuvenation action share the same common potential mechanism, which is the dilution of damage.
Concluding, we find on the one hand anti-aging molecules that are found in omics quantities within our South Atlantic diet collected in all Ourensivity actions, and on the other hand, we have theories of rejuvenation already proven in vitro. Longevity interventions, Although they do not serve to cover biological age, they do serve to attenuate certain age-related characteristics such as the reduced presence of senescent cells, and the increase in the size and functionality of the set of stem cells.
Ourense has this ability to reduce senescent cells and improve the functionality of stem cells within an epigenetic context, due to its environment, its purity of air and water, but also thanks to its diet based on indigenous products and seasonal and its traditions and healthy habits such as the presence of organic gardens, thermalism or the important sociability in traditional festivals that are deeply rooted.
The future of research takes the direction of improving the repair of damaged tissue and the wealth that Ourense has are decisive.
Pablo Gª Vivanco
Community Pharmacist
Dietician-Nutritionist.
Optician-Optometrist ES teacher on leave
Bibliografía:
Zhang, B., Trapp, A., Kerepesi, C., & Gladyshev, V. N. (2022). Emerging rejuvenation strategies—Reducing the biological age. Aging Cell, 21, e13538. https://doi. org/10.1111/acel.13538
Galkin F, Zhang B, Dmitriev SE, Gladyshev VN. Reversibility of irreversible aging. Ageing Res Rev. 2019 Jan;49:104-114. doi: 10.1016/j.arr.2018.11.008. Epub 2018 Dec 1. PMID: 30513346.Disponible en: https://pubmed.ncbi.nlm.nih.gov/30513346/
Nuñez, J. K., Chen, J., Pommier, G. C., Cogan, J. Z., Replogle, J. M., Adriaens, C., Ramadoss, G. N., Shi, Q., Hung, K. L., Samelson, A. J., Pogson, A. N., Kim, J. Y. S., Chung, A., Leonetti, M. D., Chang, H. Y., Kampmann, M., Bernstein, B. E., Hovestadt, V., Gilbert, L. A., & Weissman, J. S. (2021). Genome-wide programmable transcriptional memory by CRISPR-based epigenome editing. Cell, 184(9), 2503–2519 e2517. https://doi.org/10.1016/j.cell.2021.03.025
Petkovich, D. A., Podolskiy, D. I., Lobanov, A. V., Lee, S. G., Miller, R. A., & Gladyshev, V. N. (2017). Using DNA methylation profiling to evaluate biological age and longevity interventions. Cell Metabolism, 25(4), 954–960 e956. https://doi.org/10.1016/j.cmet.2017.03.016